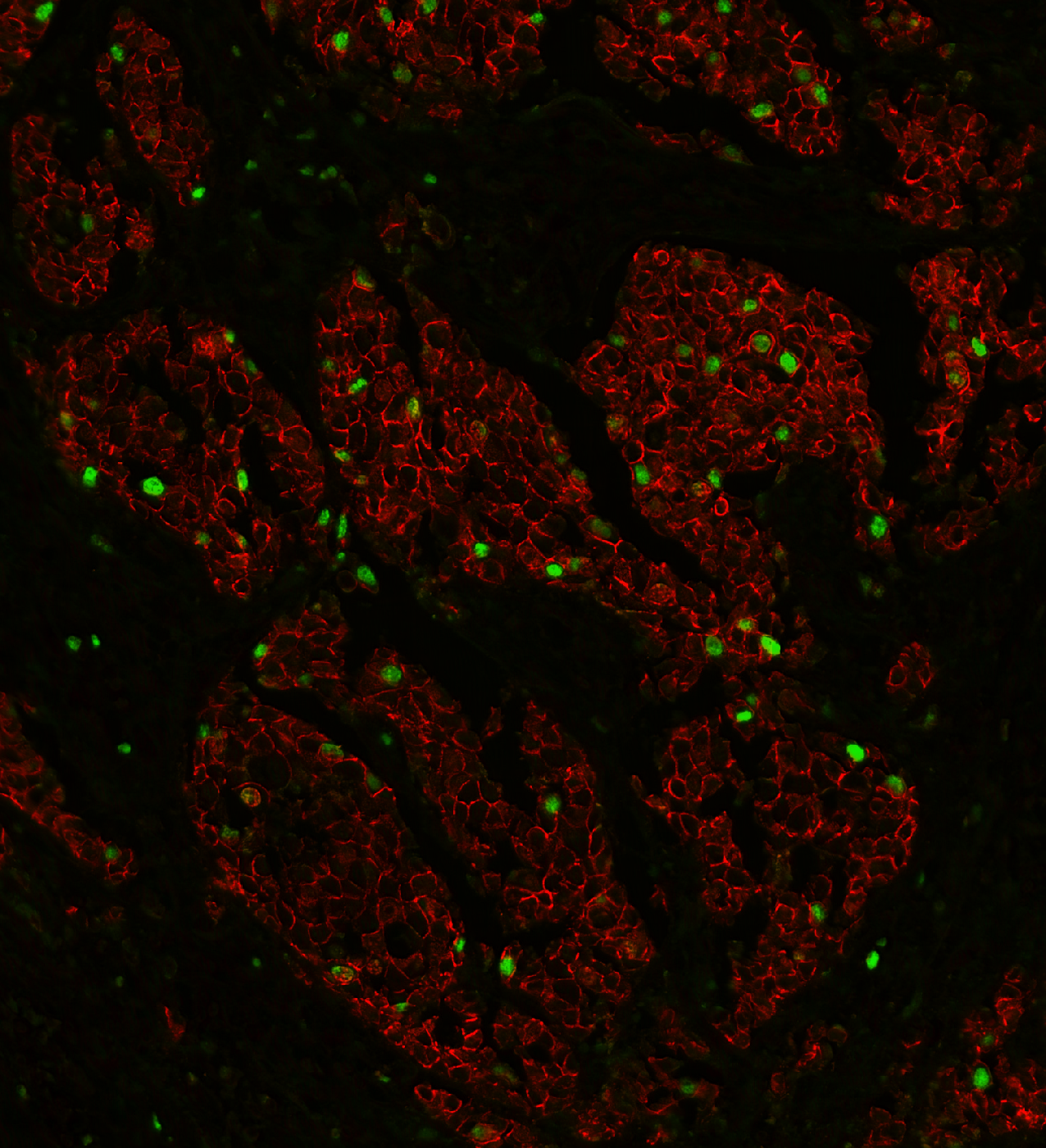
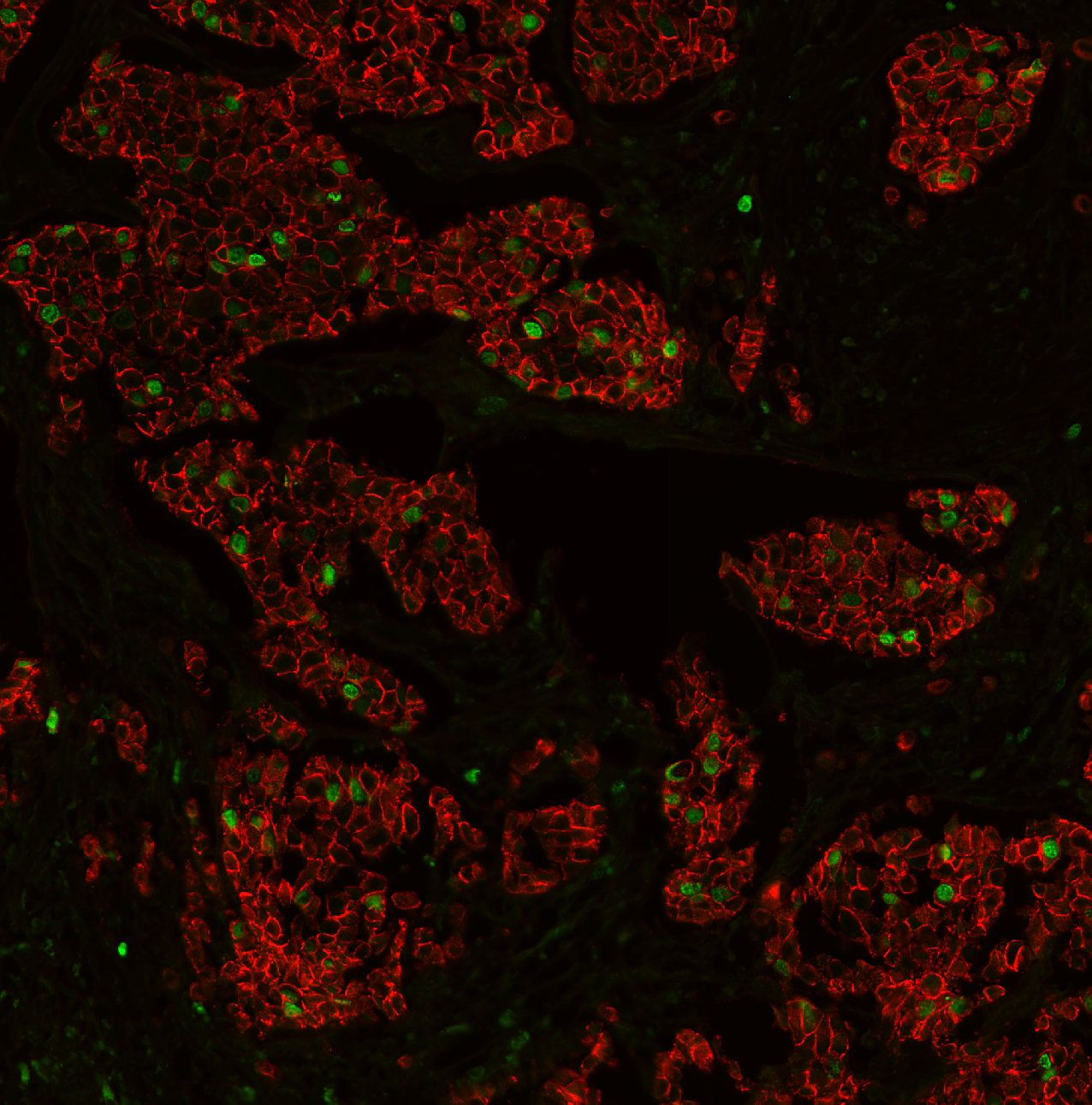
Sarpeda Plex — Marker Separation
Breast tissue: E-cadherin and Ki-67 labeled with the same fluorophore, then separated by AI. Green nuclei (Ki-67) clearly emerge from the red membrane staining (E-cadherin).
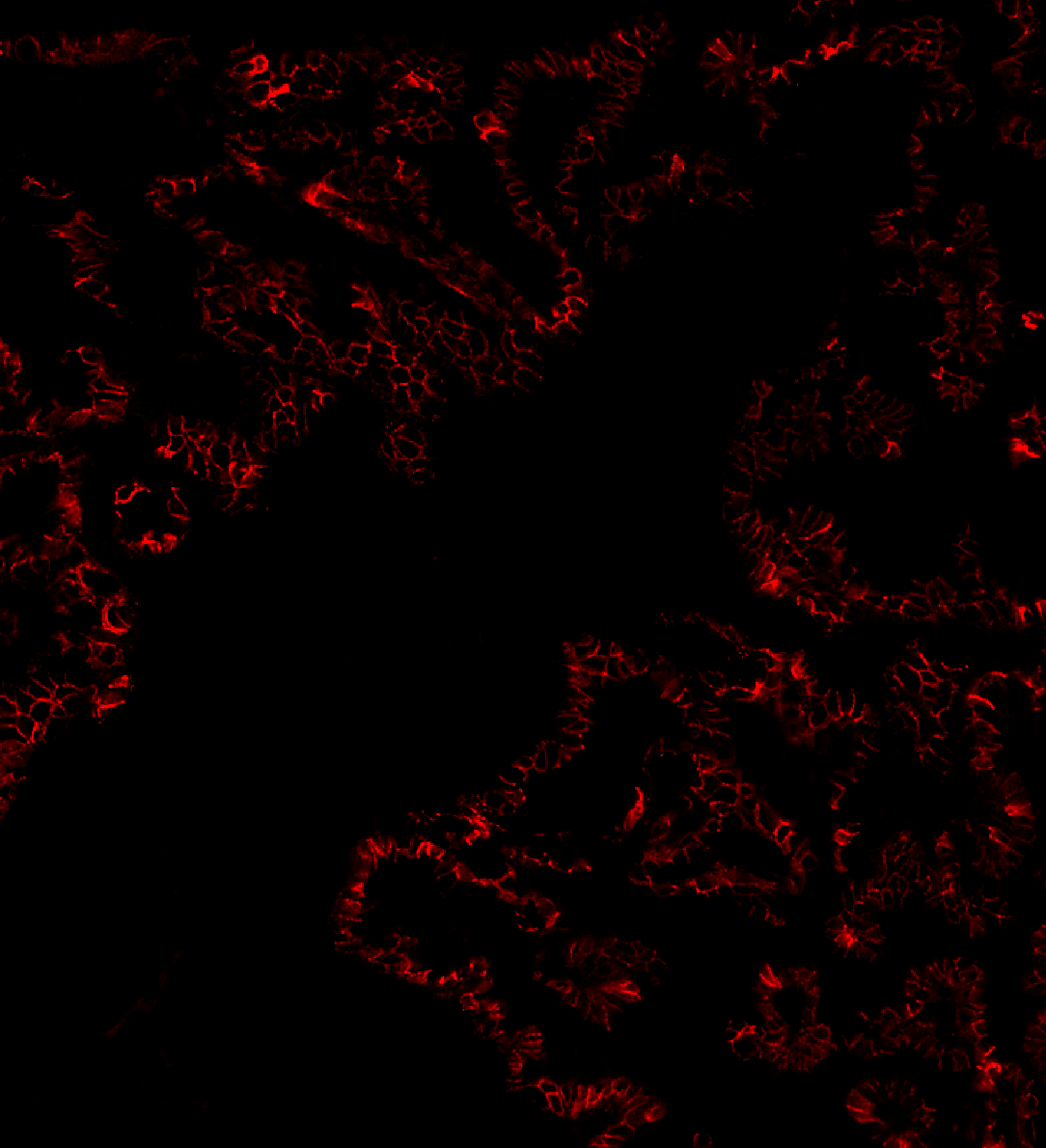
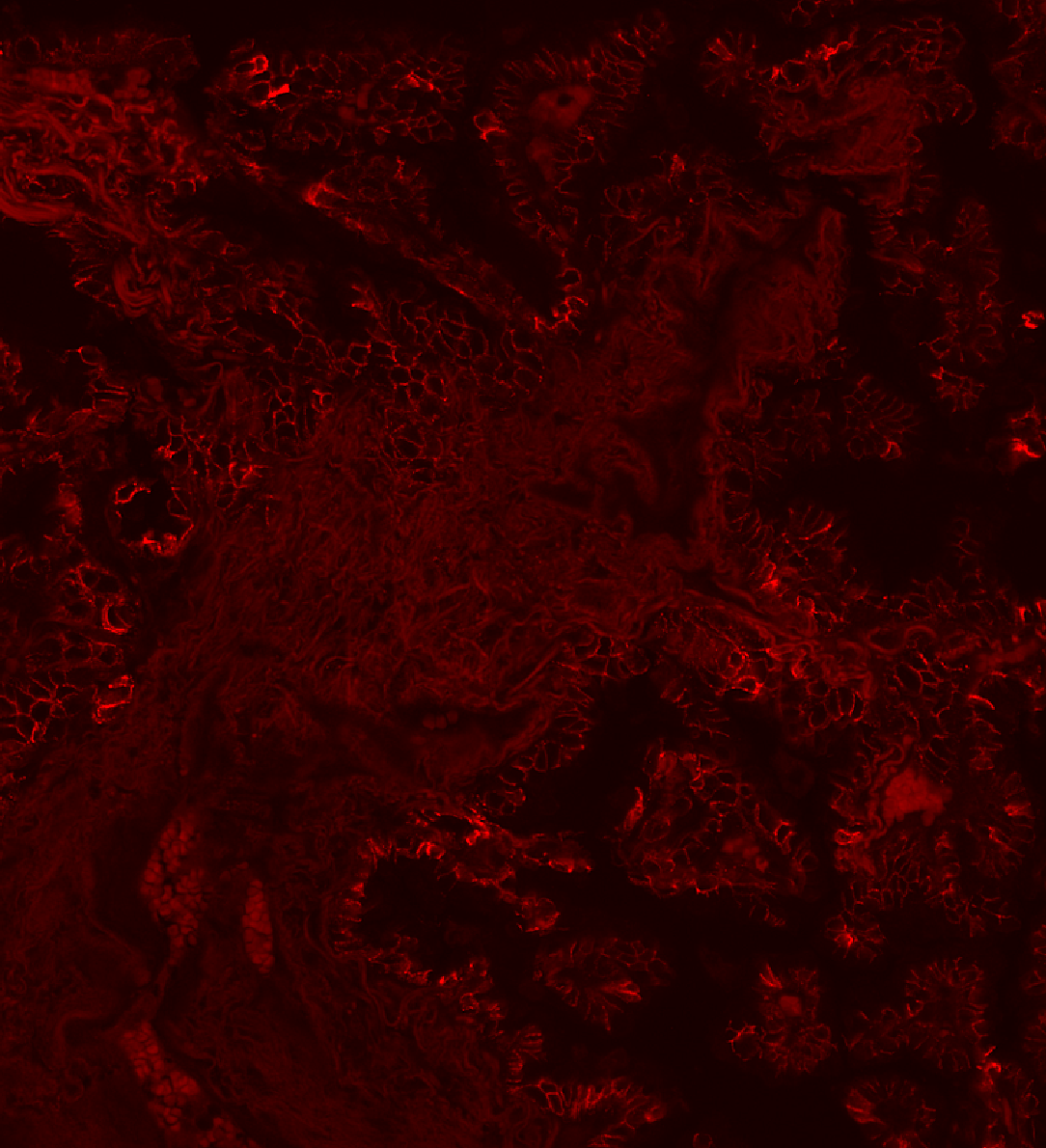
Sarpeda Clear — Background Removal
Lung tissue: E-cadherin signal buried in autofluorescence, then cleaned by AI. The true biological signal emerges with single-pixel detail preserved.